HealthPRO Canada News
May 12, 2021
A coordinated effort: How HealthPRO and Health Canada minimized the impact of drug shortages during the pandemic
Early in the pandemic, supply chain interruptions and increased demand for critical drugs around the world posed a serious threat to our drug supply. Speaking at this year’s Canadian Society of Hospital Pharmacists (CSHP) conference, HealthPRO and Health Canada described the extraordinary steps they took to ensure that didn’t happen.
___________________________________________________
Christine Donaldson, Vice President of Pharmacy at HealthPRO and Stephanie Di Trapani, A/Director, Drug Shortages Division at Health Canada are not strangers to drug shortages but the one that unfolded during the pandemic was a different kind of crisis. The situation was not just far-reaching and complex but shifted hour by hour and day to day. They knew that millions of Canadians were depending on their organizations’ ability to procure essential medications.
As we mark a year of grim statistics and staggering loss, their story of how a vast network of stakeholders, including many Canadian hospitals, responded quickly and appropriately to this crisis presented at this year’s CSHP virtual conference, is something all Canadians can be proud of.
“Many of you faced challenges and continue to face challenges, including COVID fatigue,” said Christine in her opening, “but our goal today is to share our best practices and how we plan to be in a better situation next time by strengthening our supply chain.”
One of the ways the two organizations were able to create a strong response was through the Multi-Stakeholder Steering Committee on Drug Shortages. As Stephanie described it, it’s a forum for stakeholders within the drug supply chain and the healthcare system to collaboratively improve and provide strategic guidance on the communication, mitigation, and prevention of drug shortages. “So, you can imagine that this has been an active entity throughout the pandemic.”
I - The challenges
“Occurrence is stable but persistence is going up”
Mandatory drug shortage reporting came into force in 2017 to help Health Canada harness the analytics around drug shortages. After four years of watching the numbers, they’ve noticed a disturbing trend – while the total number of shortages is steady, the shortages are lasting a lot longer. “Occurrence is relatively stable, persistence is going up,” said Stephanie.
“Right now, I can tell you that Canada is experiencing 945 unresolved shortages, and those are of the marketed drugs in Canada. This represents about 9% of those that are marketed.”
Not surprisingly, the pandemic also exposed some of the fault lines in our healthcare supply chain. “Our just-in-time supply did not serve us well during the pandemic,” said Christine. “We did not have the right products on our shelves or in our distribution systems.” Efforts to coordinate a national response were hampered by the lack of a centralized decision-making process – with no Canada-wide platform, individual provinces and territories hold most of the responsibility for the delivery of healthcare.
II - The response
“These orders had the effect of improving oversight”
When it comes to drug supplies, we operate in a global market and size matters. Canada, at only 2% of that global market, is a small player with limited volumes to offer and compete.
As demand around the world intensified for limited supply, many of the primary suppliers of injectable drugs were under pressure to sell products made for Canada to other countries and COVID hotspots. Both Health Canada and HealthPRO knew they had to work quickly to prevent that from happening.
When they realized that normal pathways of utilizing enforcement discretion was not a sustainable way of securing access to foreign supplies or resolving acute shortage situations, Health Canada enacted numerous interim orders. “These orders had the effect of improving oversight and also created a smooth pathway for needed products to come in, in a timely fashion,” said Stephanie.
National aggregation was critical
HealthPRO saw that protecting inventory meant committing to national volumes. “We worked very closely with our key hospital members to put forward commitments and build forecasts, and their volumes were used to secure the next shipments coming from overseas,” she said. She noted this was an unusual move to guarantee volumes but an essential one to protect Canadian quantities. “We’re very proud of how stakeholders and member hospitals really pulled together in a time of crisis and joined hands to meet this crisis head-on.”

Well before the pandemic, HealthPRO had taken steps to evolve its contract strategies in response to persistent drug shortages. Christine described two strategies that have mitigated supply disruptions:
- Split Awards, where the contract is awarded to not one but two suppliers, so another source is readily available if a supply disruption occurs. “Over the past few years, this strategy has mitigated over 80% of the supply disruptions.”
- Open Market strategy, a contract process that gives suppliers who want to bring in a new or alternative product to a sole source drug access to a portion of the market.
Constructing a toolbox for the future
The ground was shifting under their feet at a dizzying rate, and even as HealthPRO and Health Canada took steps to ensure a pipeline of essential supply during the pandemic, they were also thinking long-term. How could we make sure these tools serve us today and in the event of a future crisis?
One of those tools was a Critical Drugs List, compiled by HealthPRO in close collaboration with its hospital members. “We recognized that having this list of critical drugs was essential for our supplier partners and Health Canada to understand those drugs that needed to be fast-tracked into the country,” said Christine.
“We’ve been learning as we go and we realized we could no longer rely on historical demand to see what was ahead of us,” said Stephanie. A group from different departments across government came up with an Ethical Decision-Making Allocation Framework as a way of managing potential shortfalls fairly. “Having the framework has helped us be more disciplined about what drugs are coming in and where they need to go,” she says.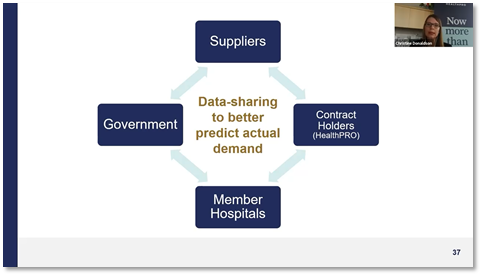
III – Opportunities for change
“Constantly looking at where we can diversify.”
As they closed their presentation, Christine and Stephanie described four ways we can be less vulnerable going forward:
#1 – Build up more supply chain resilience – Although our governments have committed to ramping up domestic production, 80-90% of our APIs (Active Pharmaceutical Ingredients) are still sourced abroad. “This is the time to build a more robust safety stock in Canada and make sure that stock is refreshed on a regular basis,” said Christine.
#2 – Greater National Coordination – In the past, efforts to deal with drug shortages were largely reactive; we’re on a new path towards greater proactivity. “We’ve ramped up surveillance and monitoring because we want to be prepared for whatever peaks and valleys the pandemic brings,” said Stephanie.
#3 – Achieve better visibility into supply chains – “One of our goals is greater transparency in all parts of the supply chain so we’re constantly looking at where we can partner and improve communication flow,” said Christine. “Another area of opportunity is better data-sharing between all stakeholders, because it's only through predictability and forecasting that we'll have a better handle on demand.”
#4 – Medium/Long-term plans for Interim Orders – As part of their strategy of having a more robust toolkit, Health Canada is considering what worked well with the interim orders and how they can be improved so they continue to ensure access to product that meets the needs of Canadian patients.



